New Research Opioids 2026: What’s Available and How They Work
New Research Opioids 2026: What’s Available and How They Work
In 2026, “research opioids” sit at the intersection of pharmacology, harm‑reduction debate, and tightly controlled chemical supply. These are not approved medical products; instead, they are high‑purity opioid‑derived compounds marketed strictly for in‑vitro or analytical research. Vendors often sell them as pellets, tablets, or powders, with clear disclaimers that they are not for human consumption.
Even though the term “research opioid” is neutral, the underlying chemistry is anything but low‑risk. Many of these compounds are structurally similar to powerful clinical opioids such as fentanyl, buprenorphine, or nitazenes, and can produce potent μ‑opioid receptor activation with steep dose‑response curves. Understanding what is actually available in 2026—and how it works at the receptor level—is essential for anyone interacting with these materials, even in theoretical or lab‑context discussions.
What “Research Opioids” Really Means in 2026
In practical terms, “research opioids” in 2026 usually refers to one of three categories:
-
Classical opioid scaffolds: Morphine‑, fentanyl‑, or etonitazene‑type analogs that replicate known opioid pharmacology but are sold without medical approval.
-
Partial‑agonist systems: Compounds like buprenorphine‑naloxone (Suboxone‑style) used to study opioid‑receptor kinetics and dependence mechanisms.
-
Novel or experimental opioid‑like tools: Designer molecules marketed as “research chemicals” whose full human toxicity and long‑term effects are not well documented.
These compounds are often distributed through research‑chemical vendors operating under strict disclaimers, emphasizing analytical or laboratory use only. In many countries, possession or distribution of such substances can fall under analogue acts or controlled‑substance‑paraphernalia laws, even if the vendor labels them as “not for human use.”
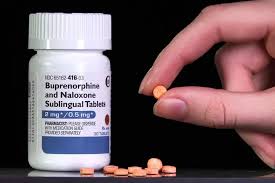
Suboxone 2mg: From Clinic to Research‑Chemical Context
One of the most recognizable research‑opioid‑style products on the market in 2026 is the “Suboxone 2mg” formulation, patterned after the FDA‑ and EMA‑approved buprenorphine‑naloxone sublingual tablet. In medical settings, Suboxone is used to treat opioid‑use disorder by reducing cravings and withdrawal while minimizing the overdose risk associated with full‑agonist opioids.
When sold as a research compound, the “Suboxone 2mg” label usually refers to a standardized blend of approximately 2 mg buprenorphine and 0.5 mg naloxone, mirroring the clinical tablet but framed for non‑therapeutic, analytical use. This makes it especially interesting for studying opioid receptor behavior, partial‑agonist dynamics, and antagonist–agonist interactions without providing a treatment‑equivalent product.
You can view the product page here (for reference only):
→ Suboxone 2mg – EURO Lab Chems
How Buprenorphine‑Naloxone Works: Mechanism and Receptor Effects
To understand modern research opioids, it helps to start with buprenorphine‑naloxone, since it illustrates several key opioid‑pharmacology concepts that generalize to many newer compounds.
Buprenorphine: A High‑Affinity Partial Agonist
Buprenorphine is a semi‑synthetic opioid that binds very tightly to the μ‑opioid receptor but activates it only partially, even at high doses. This partial‑agonist effect means:
-
It produces opioid effects (analgesia, euphoria, sedation) but with a ceiling on respiratory depression compared with full agonists like heroin or fentanyl.
-
It can displace full‑agonist opioids from receptors, reducing withdrawal and cravings in dependent individuals.
In research‑chemical form, this makes buprenorphine‑based blends useful for studying safety margins, receptor occupancy, and tolerance development in opioid‑like systems.
Naloxone: Blocking Misuse and Studying Antagonism
Naloxone is a full opioid antagonist with rapid onset when given intravenously. In the Suboxone tablet, it is included primarily to discourage injection:
-
When taken sublingually (as intended medically), naloxone has very low systemic bioavailability, so its antagonistic effect is minimal.
-
If the tablet is crushed and injected, naloxone can rapidly block opioid receptors and trigger acute withdrawal in opioid‑dependent users, acting as a deterrent.
From a research‑opioid perspective, naloxone allows scientists to model rapid opioid reversal, receptor‑occupancy competition, and the dynamics of antagonist‑agonist mixtures.
O‑DSMT and Other Research Opioid Analogs in 2026
Beyond Suboxone‑style blends, vendors also list newer opioid‑like compounds such as O‑DSMT pellets and powder, framed as “research chemicals” rather than medicines. These products are typically sold in standardized pellet formats (30 mg, 50 mg) or as loose powder, again with prominent disclaimers that they are not for human consumption and are intended solely for laboratory or analytical use.
Examples of related products (for reference only):
-
O‑DSMT pellets 50 mg – O‑DSMT Pellets 50 mg – EURO Lab Chems
-
O‑DSMT pellets 30 mg – O‑DSMT Pellets 30 mg – EURO Lab Chems
-
O‑DSMT powder – O‑DSMT Powder – EURO Lab Chems
While detailed pharmacokinetic data for O‑DSMT are scarce and often extrapolated from related opioids, the vendor‑marketed descriptions suggest it is treated as a potent μ‑opioid‑receptor agonist, analogous to other synthetic opioids that require careful dosing, analytical control, and risk‑aware handling.
Safety, Risks, and Ethical Use of Research Opioids
Because research opioids closely mimic clinical opioids, they carry many of the same risks: respiratory depression, dependence, withdrawal, and overdose—especially when combined with other CNS depressants like benzodiazepines or alcohol. In a pure research setting, these compounds are typically handled under controlled conditions, with engineering controls, fume hoods, and strict protocols to prevent accidental exposure.
Key safety considerations for research‑opioid‑style products include:
-
Potency and dosing: Even microgram‑level differences can dramatically alter effects in opioid‑like compounds, so precision analytical methods are essential.
-
Cross‑tolerance and withdrawal: Regular exposure to opioid‑like research chemicals can induce tolerance and dependence, followed by withdrawal if exposure is stopped abruptly.
-
Street‑style misuse vs. lab‑style use: When diverted into non‑research contexts, these materials become extremely hazardous, with a high risk of accidental overdose and poly‑drug interactions.
Formal medical use of opioid‑related products (like Suboxone) should always be done under the supervision of a licensed clinician, not from research‑chemical suppliers.
Why These Products Matter in Modern Research
In 2026, research opioids such as Suboxone‑style buprenorphine‑naloxone and O‑DSMT analogs are used in several legitimate scientific contexts:
-
Pharmacology studies: Probing receptor affinity, intrinsic activity, and signaling bias at μ‑, δ‑, and κ‑opioid receptors.
-
Analgesic and addiction research: Modeling pain‑pathway modulation and opioid‑use disorder interventions in cell‑based or animal models.
-
Toxicology and overdose reversal strategies: Testing naloxone and other antagonists against novel opioid analogs to refine emergency‑care protocols.
By keeping these compounds in controlled, non‑human‑use channels, researchers can generate data that ultimately inform safer clinical opioids and better harm‑reduction policies.


